Meet our scientists!
 Back
Back

Kazuhiko
Namekata
Kazuhiko
Namekata
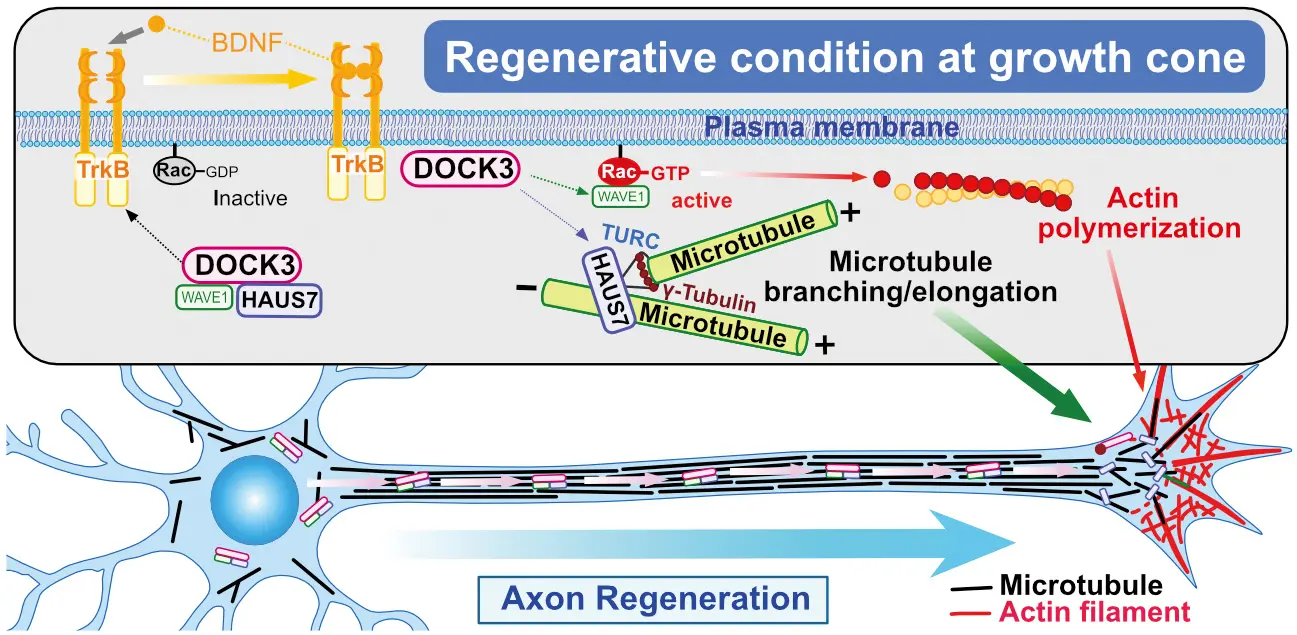
Neurons are highly efficient at transmitting information through axonal processes that can extend over long distances. However, because neurons are highly specialized post-mitotic cells, damage to neurons or their axons is difficult to repair. Dr. Kazuhiko Namekata and colleagues at the Visual Research Project study axonal regeneration using the optic nerve as a model system. Previously, they identified several key factors important for axonal regeneration following injury. They showed that constitutive activation of the BDNF/TrkB signaling pathway enhances regeneration, and further identified activators of DOCK3, a downstream target of BDNF/TrkB, that also enhance regeneration. This past year, they published a paper in Science Advances entitled “Role of HAUS7 as a DOCK3 binding partner in facilitating axon regeneration.” In this study, they build on their previous work to present a more coherent model of the steps involved in axonal regeneration. We spoke to Dr. Namekata about this work.
Could you give us some background on your DOCK3 work?
I’ve been interested in diseases associated with aging since I was a college student, and I’ve been studying DOCK3 for over 15 years because it is a protein associated with Alzheimer’s disease. The DOCK proteins are a family of intracellular signaling molecules that activate Rho family GTPases to regulate processes such as cytoskeleton remodeling, cell migration, and neurite and axonal growth and regeneration. I’ve been studying DOCK3 in particular because it is expressed specifically in neurons, and we previously found that if we overexpress DOCK3 in retinal ganglion cells (the cells whose axons compose the optic nerve), we can enhance optic nerve regeneration [after damage]. We also screened for activators of DOCK3 and identified two molecules that enhance axonal regeneration when injected into the eye.
How does the BDNF/TrkB signaling pathway relate to DOCK3?
BDNF is a secreted extracellular signaling factor that regulates processes such as neuronal survival and differentiation. It is also important for axonal regeneration. It functions by binding to and activating the cell surface TrkB receptor, which is expressed on neurons. We previously found that expressing constitutively active TrkB in retinal ganglion cells enhances optic nerve regeneration. DOCK3 is a key mediator of TrkB-dependent axonal regeneration. So, a biochemical pathway that stimulates axonal regeneration after damage would be BDNF activating TrkB, resulting in activation of DOCK3 and Rac1, which increases actin cytoskeleton remodeling required for regeneration.
How does your current paper continue this story?
For axons to regenerate, you need microtubules to polymerize, but we don’t have a connection to microtubules in our BDNF/TrkB/DOCK3 pathway. This current project started from the idea that DOCK3 is a large protein with multiple functions and multiple binding partners. We thought that there must be other important proteins that bind to DOCK3, and we identified HAUS7 as a novel DOCK3 binding partner. This is important because HAUS7 is a subunit of the augmin complex, and the augmin complex is important for microtubule branching. Without the augmin complex, branching doesn’t occur. Microtubules are critical cytoskeletal components of axons, but axons aren’t composed simply of single, straight, unbranched microtubules. Microtubules are thought to elongate through polymerization and branching nucleation. So, if you are unable to branch, you should be unable to make the long microtubule structures needed to regenerate axons.
I envisioned axons as long graceful structures which contained long, graceful, straight, unbranched microtubules.
That was the model for a long time. But, if you think about it, if all microtubules were ultra-long, unbranched structures, that could be too restricting. If there were some problem at some place in the microtubule, you wouldn’t be able to use it. On the other hand, if microtubules can branch and branches can extend, you have much more freedom. Also, if you have only long microtubules, do they have to last throughout your lifetime? Branching might allow you to repair different portions of the structure as they are damaged or get old.
What do you think is most significant about your recent paper?
I think our work is important because it links three processes, extracellular signaling from BDNF to actin skeleton remodeling through DOCK3 and Rac1 to microtubule extension and remodeling through HAUS7 and the augmin complex. Currently, our model is that HAUS7 and another protein, WAVE1, bind to DOCK3 and all three proteins are transported along microtubules to the growth cone at the end of regenerating axons. BDNF activation of TrkB induces the HAUS7-WAVE1-DOCK3 complex to move to the cell membrane at the growth cone where DOCK3 is phosphorylated. Phosphorylation of DOCK3 causes WAVE1 and HAUS7 to dissociate from DOCK3, freeing them to induce actin polymerization and microtubule assembly required for axonal regeneration.
Do you think there are any medical applications to your findings? What are your future plans?
The results from this study don’t directly translate to medical treatments, since we didn’t increase regeneration. Instead, we identified mechanisms that are important for regeneration. However, I think our future studies may be more relevant to medicine. In this study, we made Haus7 knockout mice [mice in which Haus7 is deleted]. Surprisingly, these mice are alive, and HAUS7 isn’t a completely essential gene. It’s possible that HAUS7 may not be an essential subunit of the augmin complex or that other microtubule nucleation pathways can compensate for augmin deficiencies. What we do know is that Haus7 knockout mice are alive but are deficient for axonal regeneration. We also know that Haus7 knockouts are defective for processes that require rapid cell proliferation, since cell division requires microtubules and microtubule branching. So, Haus7 KO mice are defective for processes such as spermatogenesis, thymic function, and microglial proliferation. Since immune responses require rapid responses and rapid proliferation of immune cells, I believe that inhibiting HAUS7 activity or augmin function might be a viable strategy for combating autoimmune diseases such as multiple sclerosis (MS), and I’m interested in pursuing these types of studies in the future. This strategy might also work as a possible cancer treatment, although I’m not actively pursuing that avenue now. Finally, I think that inhibiting HAUS7 activity might accelerate the onset and severity of diseases such as Alzheimer’s disease and glaucoma. So, I believe that HAUS7, augmin, and altered microtubule structures could contribute to various diseases and I’m eager to study these connections in the future.
Interviewed by Jun Horiuch