
Misfolding and aggregation of normally soluble proteins are common pathological features of many neurodegenerative diseases, including Alzheimer’s, Parkinson’s, Creutzfeldt–Jacob and Huntington’s diseases. For example, Parkinson’s disease (PD), dementia with Lewy bodies (DLB) and multiple system atrophy (MSA) are characterized by accumulation of misfolded α-synuclein in neuronal and/or glial cells, and therefore these diseases are termed α-synucleinopathies. In PD and DLB, α-synuclein pathologies are mainly observed in neurons in the form of Lewy bodies and Lewy neurites, while glial cytoplasmic inclusions are seen in oligodendrocytes in MSA. Previous studies have suggested that α synuclein has the properties of prion, seed dependent aggregation, cell to cell propagation and the existence of distinct aggregate conformations (strains). There is a relationship between protein aggregate conformation and clinical phenotype in prion diseases, however, whether differences in the strains of α synuclein aggregates account for the different pathologies remained unclear.
In this study, we prepared recombinant α-synuclein monomer, agitated it in the presence or absence of salt at a physiological concentration and generated two types of α-synuclein fibrils, α-synuclein fibrils (+) and α-synuclein fibrils (-), from identical monomer. We injected α-synuclein fibrils (+) and α-synuclein fibrils (-) into striatum of wild-type mice, and after one month, we examined the accumulation of phosphorylated α synuclein deposits resembling those observed in patients’ brains. We found that α-synuclein fibrils (-) induced Lewy body/Lewy neurite-like abnormal phosphorylated α-synuclein deposits through the mouse brain, whereas few phosphorylated α-synuclein deposits were induced by α-synuclein fibrils (+) (Figure 1). We also investigated their seeding and propagation ability in mice primary-cultured neurons and found that α-synuclein fibrils (-) induced marked accumulation of phosphorylated α-synuclein and ubiquitinated protein aggregates, while α-synuclein fibrils (+) did not. We found that onlyα-synuclein fibrils (-) apparently inhibited proteasome activity in vitro and co-precipitated with purified 26S proteasome complex. Further examination indicated that structural differences in the C-terminal region of α synuclein fibrils lead to different effects on proteasome activity. These results provide a possible molecular mechanism to account for the different pathologies induced by different α synuclein strains (Figure 2). Overall, we found that the degree of interaction with proteasome and the inhibition of proteasome activity vary depending on the strain of α-synuclein aggregates. The relationship between proteasome inhibition and the induction of the pathology supports the hypothesis that prion-like activity of α-synuclein aggregates contributes to disease progression.
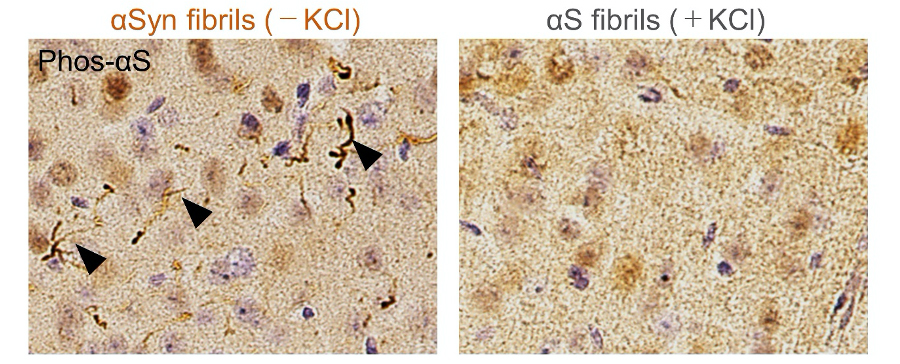
Comparison of pathologies in WT mouse brains by inoculation of two α-synuclein strains. Distribution of phosphorylated α-synuclein pathology in mouse brain. α-synuclein fibrils (-) or α-synuclein fibrils (+) were injected into the striatum of WT mouse brain and stained with phosphorylated α-synuclein antibody 1 month after injection. Typical pathological α-synuclein deposits are indicated by the arrowheads. Sections were counterstained with hematoxylin. Representative images of cortex of mice injected with α-synuclein fibrils (-) (left) or α-synuclein fibrils (+) (right) are shown.
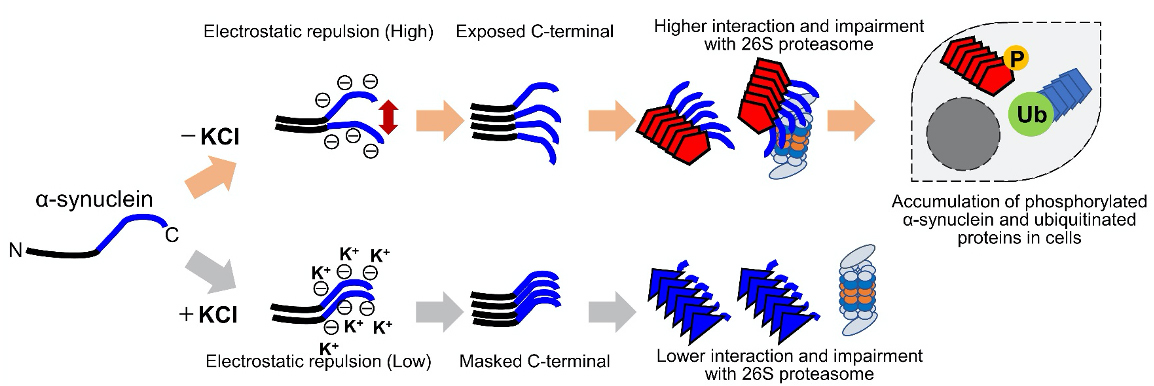
Schematic representation of α-synuclein strain formation and strain dependent interaction with 26S proteasome.
In the absence of salt, α-synuclein monomers have exposed C-terminal region with high electric repulsion. These form the α-synuclein strain with exposed C-terminal region and this type of the α-synuclein strain can interact with 26S proteasomes and inhibit their activities resulting the accumulation of phosphorylated α-synuclein aggregates and ubiquitinated proteins (left). In the presence of salt, α-synuclein monomers have packed C-terminal region with low electric repulsion. These form the α-synuclein strain with masked C-terminal region and this type of the α-synuclein strain can not interact with 26S proteasomes.