A research group led by Motomi Matsuno of the Higher Brain Functions Project and Junjiro Horiuchi of the Academic Support Office at our institute has revealed that age-related memory decline is caused not by “forgetting,” but by “memory confusion” (generalization), in which distinct memories become mixed.
They further found that this phenomenon is driven by excessive activity of dopaminergic neurons during the process of memory consolidation. This discovery may lead to a new understanding of age-related memory disorders and the development of novel therapeutic approaches.

As humans and other animals age, their memory decreases. This age-related memory impairment may be thought to occur through an increase in forgetting or a gradual loss of memory. However, in this study, the authors found that a major cause of age-related memory impairment is increased uncertainty about a memory. Aged animals make inappropriate memory connections and recall memories at inappropriate times leading to confusion. Inappropriate memory connections are formed in old animals because they are unable to decrease dopamine signaling during memory consolidation.
Memory decreases upon aging in many organisms including humans and Drosophila. However, the precise causes of this decrease have not been completely elucidated. Memory is thought to be encoded in the brain in specific neuronal networks that are formed during learning or training of an association. For example, during formation of long-term memories, certain neurons become active and are referred to as memory engram cells. Inhibiting activity of memory engram cells prevents a memory from being recalled, while artificial activation of engram cells seems to induce memory recall even in the absence of memory cues. These results demonstrate that memory engram cells are important for long-term memories. Since aging causes a decrease in long-term memories, the authors hypothesized that aging may affect the formation or activity of memory engram cells.
Drosophila can be electrically shocked multiple times while being exposed to an odor. After this experience, they learn to associate the odor with pain and subsequently avoid the odor. If flies are trained in this association multiple times with appropriate rests in between trainings, they form long-term memories of this association. Similar to other animals, long-term memory in flies depends on memory engram cells. The authors decided to examine whether engram cells were formed and behaved normally in old flies.
In this study, the authors found that similar numbers of engram cells are produced in both young and old flies during the formation of long-term memories. Activation of these engram cells induces avoidance behaviors in both young and old flies, demonstrating that they are functional. However, while engram cells in young flies are activated specifically by the odor that was paired with electrical shocks during training, engram cells in old flies are activated by many different odors. This suggested that memory impairments in old flies are not caused by the loss of memories, but instead are caused by confusion resulting from the formation of inappropriate associations that interfere with proper memory recall. This phenomenon is known as aberrant memory generalization. To verify this hypothesis, the authors trained young and old flies and demonstrated that young flies avoid only the odor that was paired with electrical shocks during training, while old flies avoid many different odors.
The authors further identify a cellular pathway consisting of glutamatergic neurons, dopaminergic neurons and engram cells that regulates memory generalization. Specifically, glutamatergic activity increases dopaminergic activity which, in turn, increases plasticity of engram cells during memory consolidation. This increased plasticity allows various sensory pathways to activate memory engrams, inducing generalization and reducing memory accuracy. Supporting this model, increasing dopaminergic activity during consolidation increases generalization in young flies and decreases memory, while decreasing dopaminergic activity in old flies decreases generalization and improves memory.
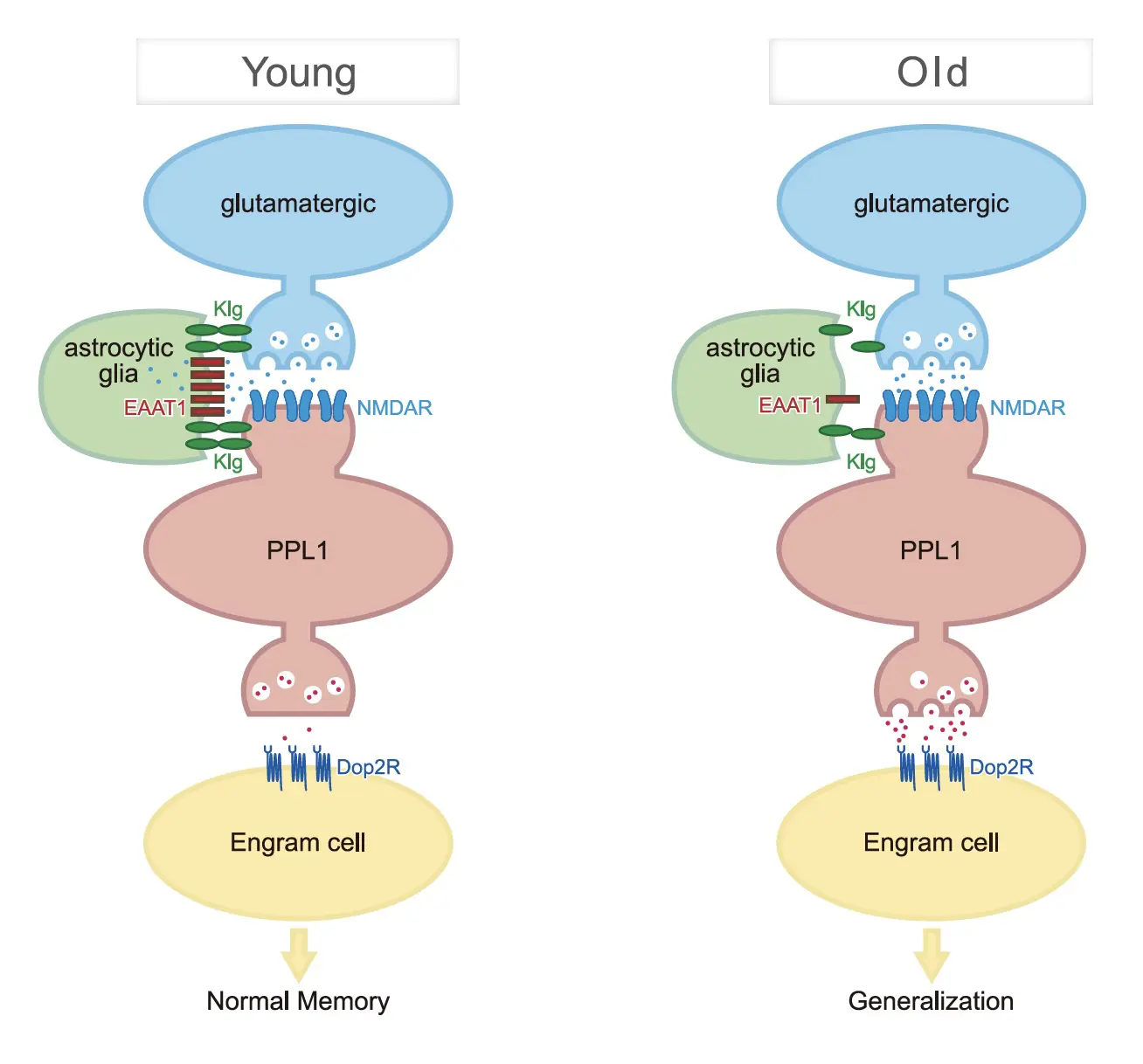
This study has revealed a novel mechanism by which age-related memory decline is driven by memory generalization, rather than simple forgetting.
Future work will examine whether this mechanism is conserved in mammals and humans, which is expected to advance our understanding of abnormal memory reactivation observed in conditions such as age-related cognitive impairment and post-traumatic stress disorder.
In addition, the finding that modulation of dopamine and glutamate signaling can restore memory specificity suggests the potential for developing new therapeutic strategies for memory disorders.
This work was supported by Japan Society for Promotion of Science (JSPS) KAKENHI Grants (JP20K06753 to MM; JP19H01013 and JP21K18238 to MS; and JP21K06403 and JP18K06496 to JH).