
Narcolepsy type 1 (NT1) is a sleep disorder characterized by excessive daytime sleepiness and cataplexy. Genetic factors are associated with the development of NT1. To date, our research group has identified several fatty acid metabolism-related genes as susceptible loci for NT1. Abnormality in fatty acid metabolism has been suggested to be involved in the pathophysiology of NT1. However, the mechanisms still remain uncertain. In the present study, we conducted metabolome analysis using cerebrospinal fluid (CSF) from patients with NT1 to discover the molecular basis of NT1 in the brain.
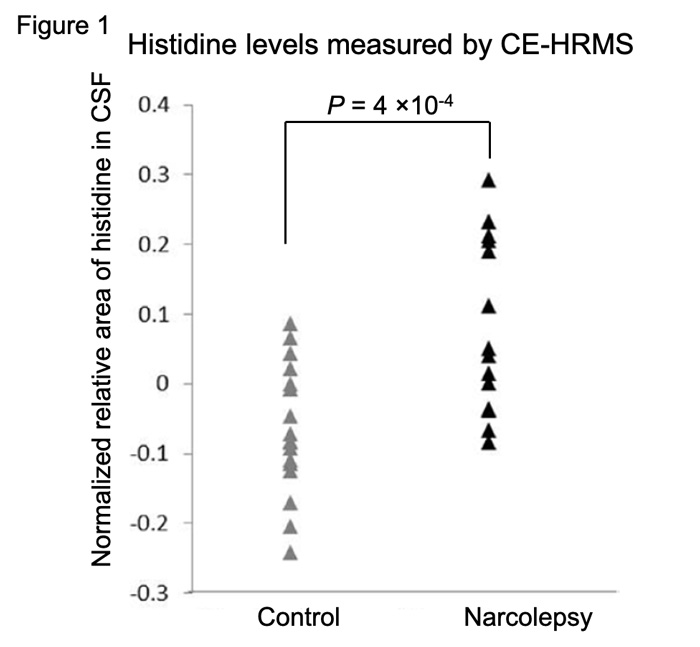
Metabolome analysis of CSF is challenging due to the relatively low levels of metabolites in CSF. In the present study, metabolites in CSF from 14 patients with NT1 and 17 control subjects were examined with a new technology, capillary electrophoresis coupled with Fourier transform mass spectrometry (CE-FTMS). A total of 268 metabolites were successfully detected. The detected metabolites were compared between NT1 and control groups. As a result, levels of histidine which is an essential amino acid was the most significantly increased in patients with NT1 (P = 4 × 10–4) (Figure 1). We next investigated whether metabolites with nominally significant associations were enriched in specific pathways. Glycine, serine, and threonine metabolism was detected as the most significant pathway. Other significant pathways included alanine, aspartate, and glutamate metabolism. Interestingly, these amino acids associated with NT1 were mainly glycogenic amino acids and were increased in patients with NT1. Our previous studies have revealed that dysfunction in fatty acid metabolism was associated with NT1. Therefore, the results imply that glycogenesis may be enhanced in NT1 as a compensatory mechanism for a lower activity of fatty acid metabolism.
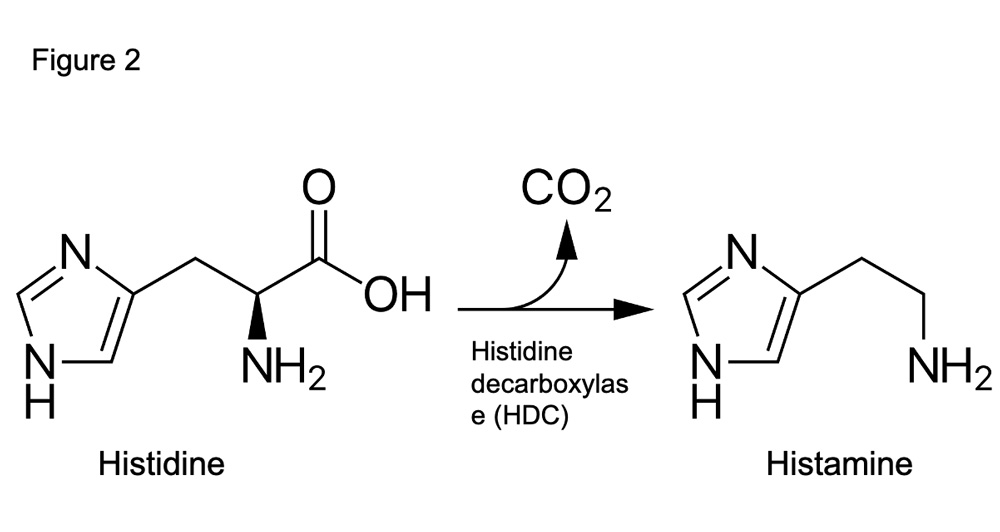
Histidine is converted to histamine by histidine decarboxylase (HDC) (Figure 2). Histamine is a neuromodulator involved in not only inflammatory and allergic responses but also the regulation of sleep-wakefulness. Antihistamines which are medicines used to reduce allergy symptoms such as sneezing, itching and rashes can cause drowsiness as a side effect, due to their ability to block histaminergic systems in the brain. In addition, the first-generation antihistamines are available over the counter as sleep aids.
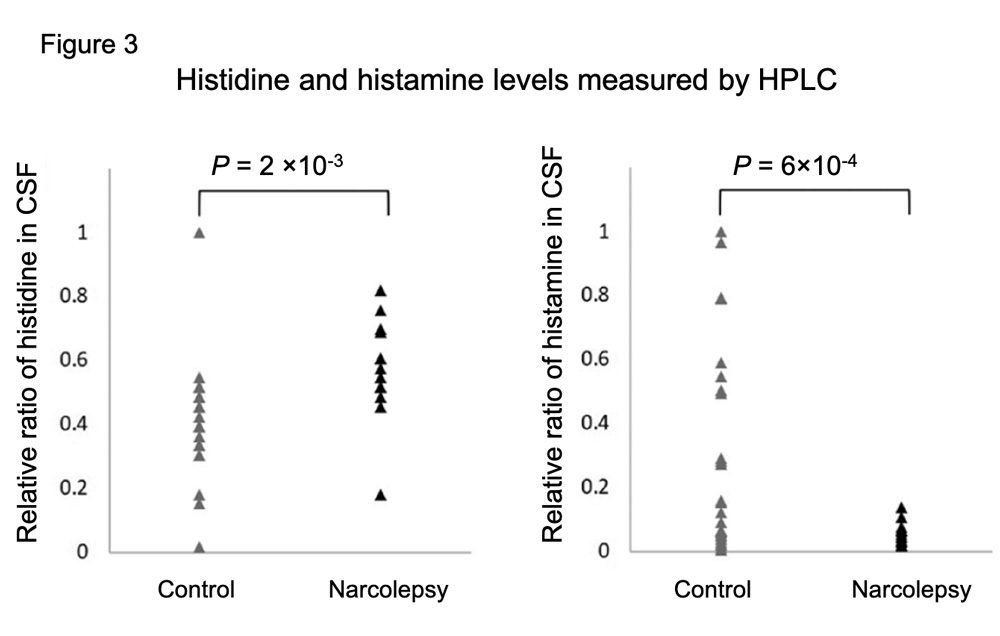
Therefore, we examined levels of histamine and histidine in CSF using high-performance liquid chromatography (HPLC). With newly recruited additional samples (4 patients with NT1 and 11 control subjects), a total of 18 patients with NT1 and 28 control subjects were analyzed for the HPLC measurement. As a result, histidine levels in NT1 patients were significantly higher than those in control subjects (P = 2 × 10–3) (Figure 3). The increase of histidine in NT1 patients was validated by HPLC including more samples. Meanwhile, histamine levels in NT1 patients were significantly lower than those in control subjects (P = 6 × 10–4) (Figure 3). These results suggest that histidine biosynthesis is upregulated as homeostatic compensation but histidine isn’t effectively converted to histamine.
Subjects (14 patients with NT1 and 17 controls) whose metabolites of CSF were examined with CE-FTMS underwent polysomnography (PSG) and the multiple sleep latency test (MSLT). Exploratory analyses were conducted to find metabolites associated with conventional sleep variables obtained from PSG and MSLT. We found a significant association between 5’-deoxy-5’-methylthioadenosine (MTA) and apnea–hypopnea index (AHI: the number of apnea plus hypopnea events per hour of sleep). A negative correlation was shown between MTA and AHI. The functions of MTA in sleep remain unclear. Future studies are required to examine the functions of MTA in sleep apnea or hypopnea especially focusing on its anti-inflammatory effects. In addition, gamma aminobutyric acid (GABA) and caffeine metabolism were found to be correlated with rapid eye movement sleep latency (REML). Previous studies have reported that GABA and caffeine affect sleep. In this study, we confirmed that they are especially associated with the appearance of REM sleep.